-
Spin Column for DNA/Rna Extraction/Virus Extraction 8/6-Layer Silica Membrane

Basic Info. Model NO. NLD450-451
-
Quality Assurance Mascarilla Desechables Disposable Medical Grade Face Masks with Designs

Overview Product DescriptionMaterial:Our AdvantagesFAQ1. Who are we?We are based in Hubei, China, start from 2015,sell to Domestic Market(25.00%),North America(20.00%),South America(10.00%),Western Europe(10.00%),Southeast Asia(5.00%),Eastern Asia(5.00%),Northern Europe(5.00%),Sou
-
Singderm Silk New-Developed Hyaluronic Sodium Acid Dermal Filler for Lip Filler
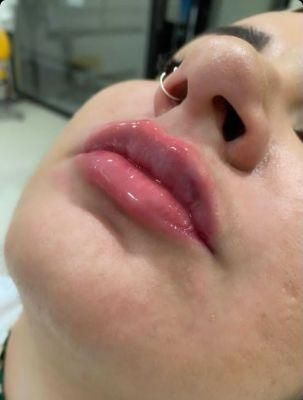
Overview Product DescriptionSpecificationOur AdvantagesApplicationsRecommended UseMain ProductsCompany Profile
-
Factory Direct Sale Ce/FDA/Approved Single or Double Medical Disposable Elastic Surgical Non Woven Cap

Basic Info. Model NO. HF CP1
-
Good Biocompatibility Anti-Wrinkle Dermal Filler with Smooth Injection and Sealing

Overview Product DescriptionOur AdvantagesApplicationsRecommended UseMain ProductsCompany Profile
-
100% Nature Cotton Medical Absorbent Non-Sterilized and Sterilized Cotton Balls

500g/bag,20bag/ctn64*46*46CMCertificationsCompany InformationPackaging&shippingFAQQ1. What is your terms of packing?A: Generally, we pack our goods in neutral brown cartons. If you have legally registered patent, we can pack the goods in your branded boxes after getting your authorization letter
-
Rapid Testing Kits Nose Test Rapid Antigen Kit 19 Test
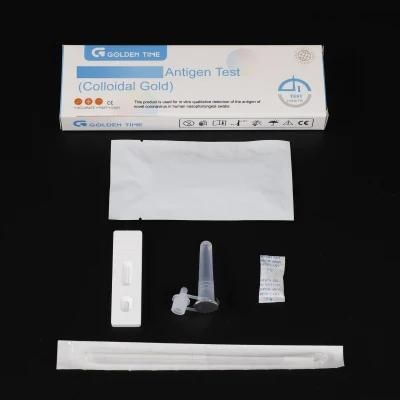
AntigenTest KitINTENDED USEThis product is used for in vitro qualitative detection of the antigen of novel virusin human nasopharyngeal swabs.FEATURES AND BENEFITS• RESPONSIVE:High accuracy and stability,double antigen sandwich assay.• QUICK CHECK:Easy to use in simple step,15 mi
-
Viraltransportation Tube Plastic Stick Nylon Nasal Flocked Swab Non-Inactivated Vtm
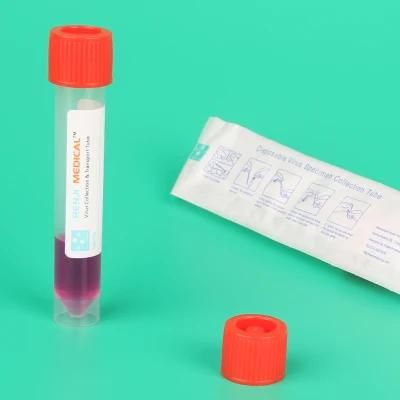
SpecificationsModel NO.SL901AProduct NameDisposable virus specimen collection tubeStandard1 swab +10ml /7ml/5ml tube+3ml inactivation /Non-inactivation medium + funnelMaterialTube:PP Cap:PEApplicationFor the Saliva collection and DNA test and Transportation of clinical virus samplesCertificationNMPA