Shenzhen Kangdaan Biological Technology Co., Ltd.
Shenzhen KangDaAn Biotechnology Co., Ltd. Located in Nanshan District, Shenzhen, is a high-tech company integrating R& D, production and sales of IVD sampling consumables and IVD equipment consumables.
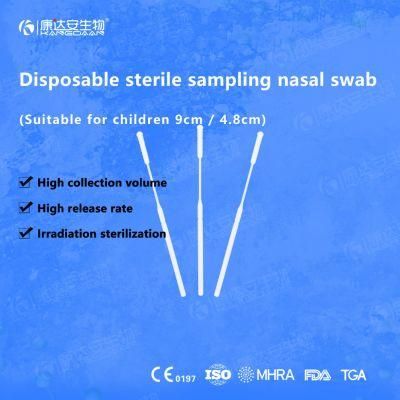
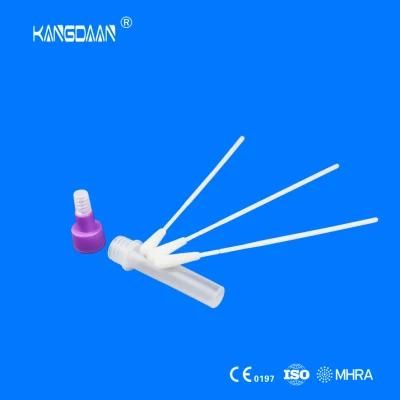
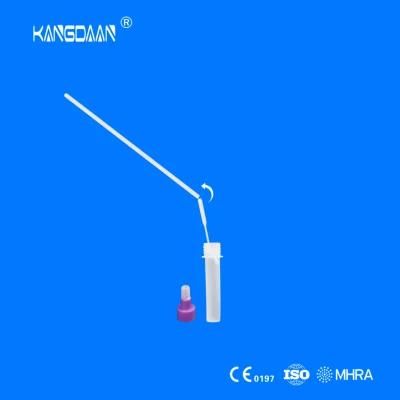
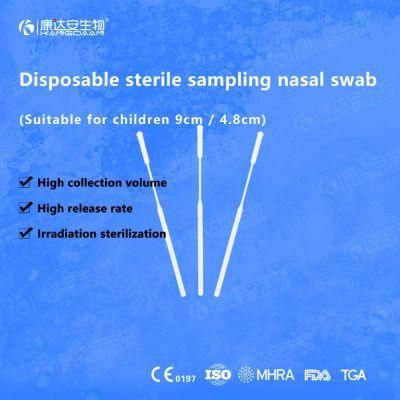
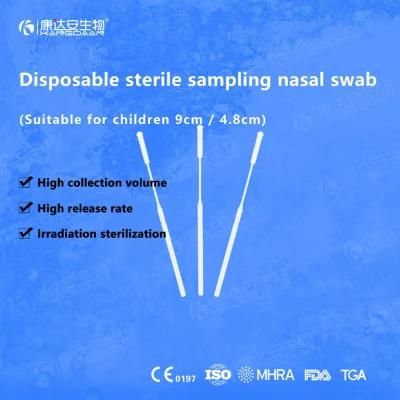
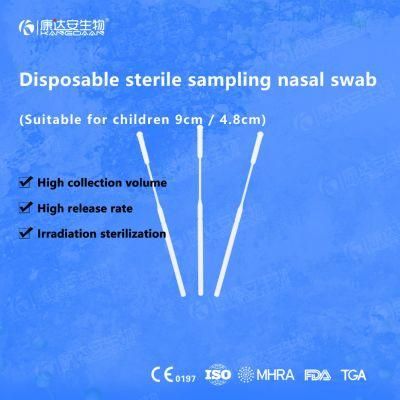
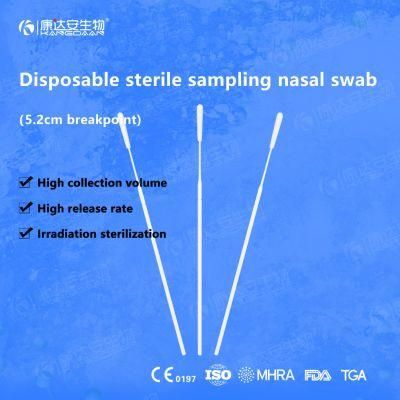
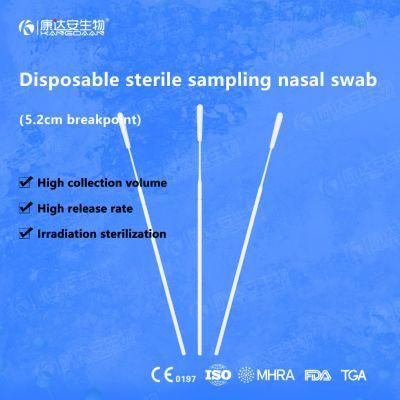
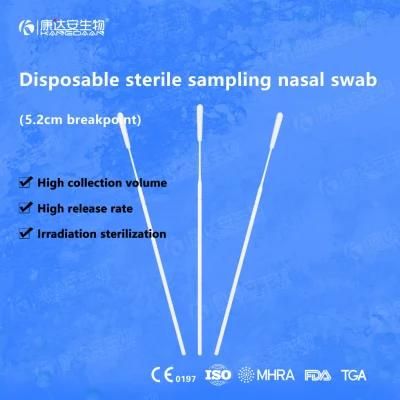
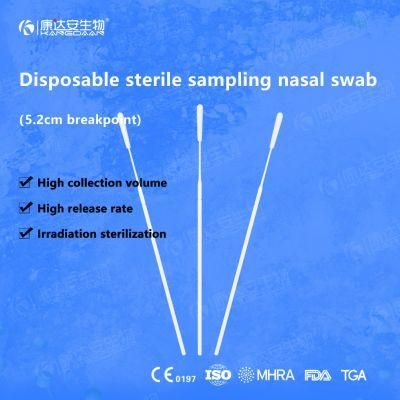
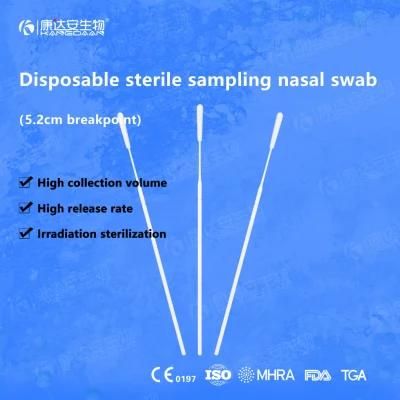
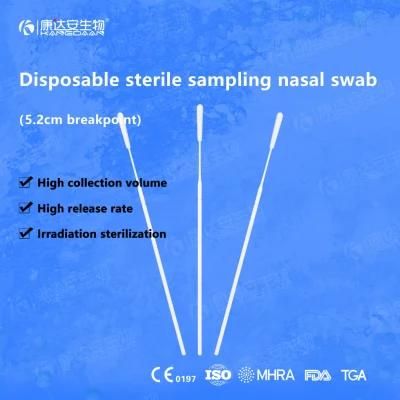
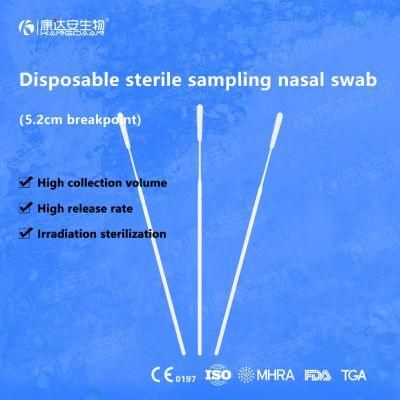
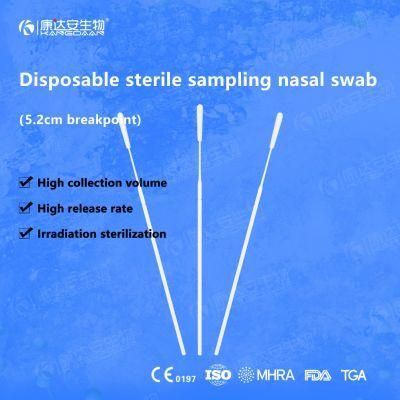
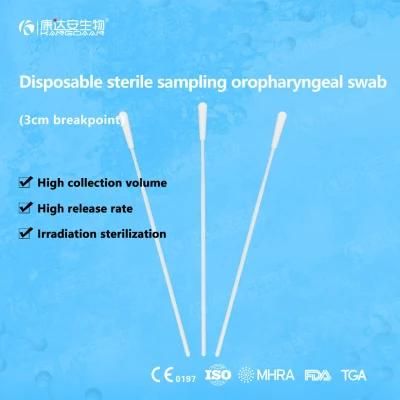
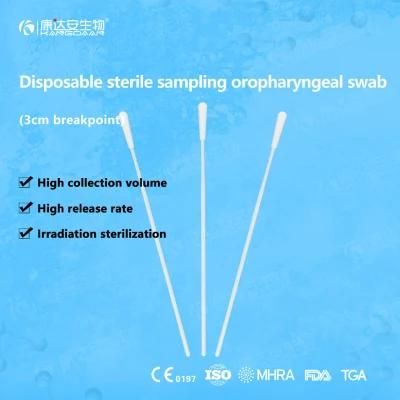
KangDaAn adheres to the idea of "talent is the first productive force of an enterprise", and has established a professional and technological team, constantly innovating products centered on IVD sampling consumables, and relying on customer needs to form a series of sampling consumables solutions, products include disposable sampling swabs( Nasopharyngeal swab, Oropharyngeal Swab, Nasal swab, Sponge Swab), disposable cervical sampling brushes, VTM, Cell preservation solution, antigen sampling solution, antibody sampling solution, etc.; At the same time, it has extended in the field of IVD, and launched IVD equipment such as biochemical analyzer, blood cell analyzer, immunofluorescence analyzer, etc. More IVD inspection consumables have been launched on the technology platform, such as biochemical reagents, colloidal gold, and PCR. The company is willing to work with partners and company employees to move forward and develop together.
KangDaAn is one of the largest sampling swab manufacturers in China in terms of production capacity and standard; Our complete supply chain includes a precision injection molding factory, flocked&sponge swab factory, 100, 000-grade automated clean workshop with 5000 sqm+ storage space. Our swabs have been well approved and listed by many authorities both at home and abroad: Class II sterile registration certificate, ISO13485 system certification, EU CE0197(MDR) announcement certification, US FDA certification, UK MHRA registration, Australia TGA, France FSC, Vietnam FSC, Singapore HSA, Thailand TFDA, and have been exported to more than 40 countries and regions around the word.
Enterprise development history
· In 2020, Obtained CE0197 certification under the new EU medical device MDR system
· In 2022, obtained Thailand TFDA certification
· In 2022, obtained Singapore HSA certification
· In 2022, obtained the free sales certificate of French FSC and Vietnam FSC
· In 2022, obtained the Australian TGA system certification
· In 2021, passed the British MHRA registration
· In 2021, the US FDA certification has been obtained
· In 2020, obtained the EU CE0197 system certification
· In 2020, passed the ISO13485: 2012 medical device production quality management system certification, completing the big leap in the development of KangDaAn biological quality
· In 2020, obtained the second-class medical device registration certificate, and completed the big leap in the development of KangDaAn′s biological quality
· In 2018, KangDaAn passed various inspection indicators and was awarded the license of medical device production enterprise.
KangDaAn adheres to the idea of "talent is the first productive force of an enterprise", and has established a professional and technological team, constantly innovating products centered on IVD sampling consumables, and relying on customer needs to form a series of sampling consumables solutions, products include disposable sampling swabs( Nasopharyngeal swab, Oropharyngeal Swab, Nasal swab, Sponge Swab), disposable cervical sampling brushes, VTM, Cell preservation solution, antigen sampling solution, antibody sampling solution, etc.; At the same time, it has extended in the field of IVD, and launched IVD equipment such as biochemical analyzer, blood cell analyzer, immunofluorescence analyzer, etc. More IVD inspection consumables have been launched on the technology platform, such as biochemical reagents, colloidal gold, and PCR. The company is willing to work with partners and company employees to move forward and develop together.
KangDaAn is one of the largest sampling swab manufacturers in China in terms of production capacity and standard; Our complete supply chain includes a precision injection molding factory, flocked&sponge swab factory, 100, 000-grade automated clean workshop with 5000 sqm+ storage space. Our swabs have been well approved and listed by many authorities both at home and abroad: Class II sterile registration certificate, ISO13485 system certification, EU CE0197(MDR) announcement certification, US FDA certification, UK MHRA registration, Australia TGA, France FSC, Vietnam FSC, Singapore HSA, Thailand TFDA, and have been exported to more than 40 countries and regions around the word.
Enterprise development history
· In 2020, Obtained CE0197 certification under the new EU medical device MDR system
· In 2022, obtained Thailand TFDA certification
· In 2022, obtained Singapore HSA certification
· In 2022, obtained the free sales certificate of French FSC and Vietnam FSC
· In 2022, obtained the Australian TGA system certification
· In 2021, passed the British MHRA registration
· In 2021, the US FDA certification has been obtained
· In 2020, obtained the EU CE0197 system certification
· In 2020, passed the ISO13485: 2012 medical device production quality management system certification, completing the big leap in the development of KangDaAn biological quality
· In 2020, obtained the second-class medical device registration certificate, and completed the big leap in the development of KangDaAn′s biological quality
· In 2018, KangDaAn passed various inspection indicators and was awarded the license of medical device production enterprise.
Product List